Risk / Compliance Guide
I need to identify and act on the active risk conditions in my industry with structured evidence.
You monitor the risk landscape for your industry continuously. You need structured, evidence-based risk frameworks that map confirmed conditions to mitigation steps — not generic risk lists.
Working example
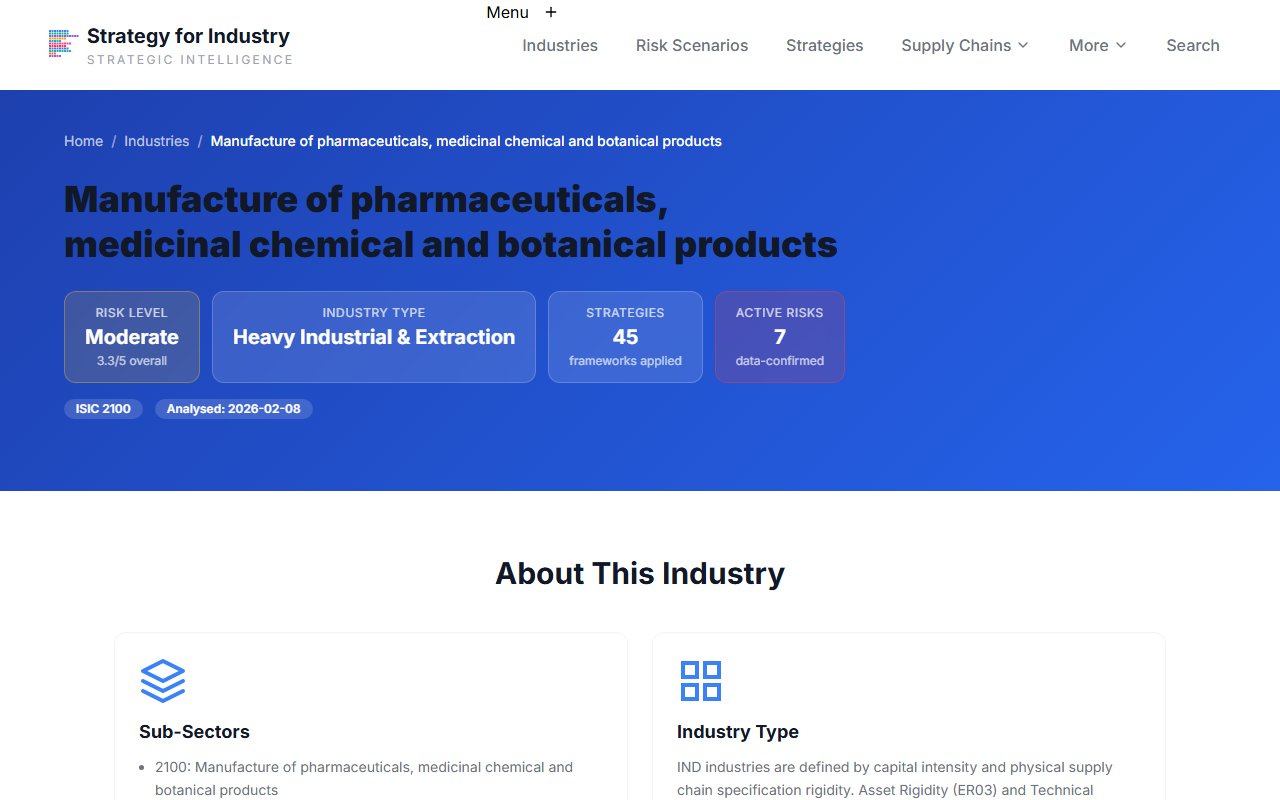
Manufacture of Pharmaceuticals ISIC 2100
New ESG supply chain compliance requirements take effect in Q3. You need to identify which risk conditions are currently confirmed for pharmaceutical manufacturing, map them to mitigation playbooks, and prepare a briefing for your executive team by end of the week.
Journey 1
Run a full structured risk review for your industry
Identify every active risk condition for your industry, review trigger conditions and business impact, and map each to a tactical mitigation response.
Industry Hub
Open your industry and review confirmed active risks
Find your industry and scroll to 'Confirmed Active Risks' on the hub page.
Confirmed active risks are score-triggered: the industry's 83-attribute scores meet the threshold conditions of each risk rule. These are not hypothetical — they're conditions the industry's profile actually satisfies.
Matched Scenario
Open a matched scenario for full risk detail
Click through to a matched scenario page for any confirmed risk.
Each matched scenario shows: exact trigger conditions (which attributes, at what score thresholds), business impact narrative, cascade risks (what triggers next if unmitigated), and the mitigation hint. This is your primary risk register input.
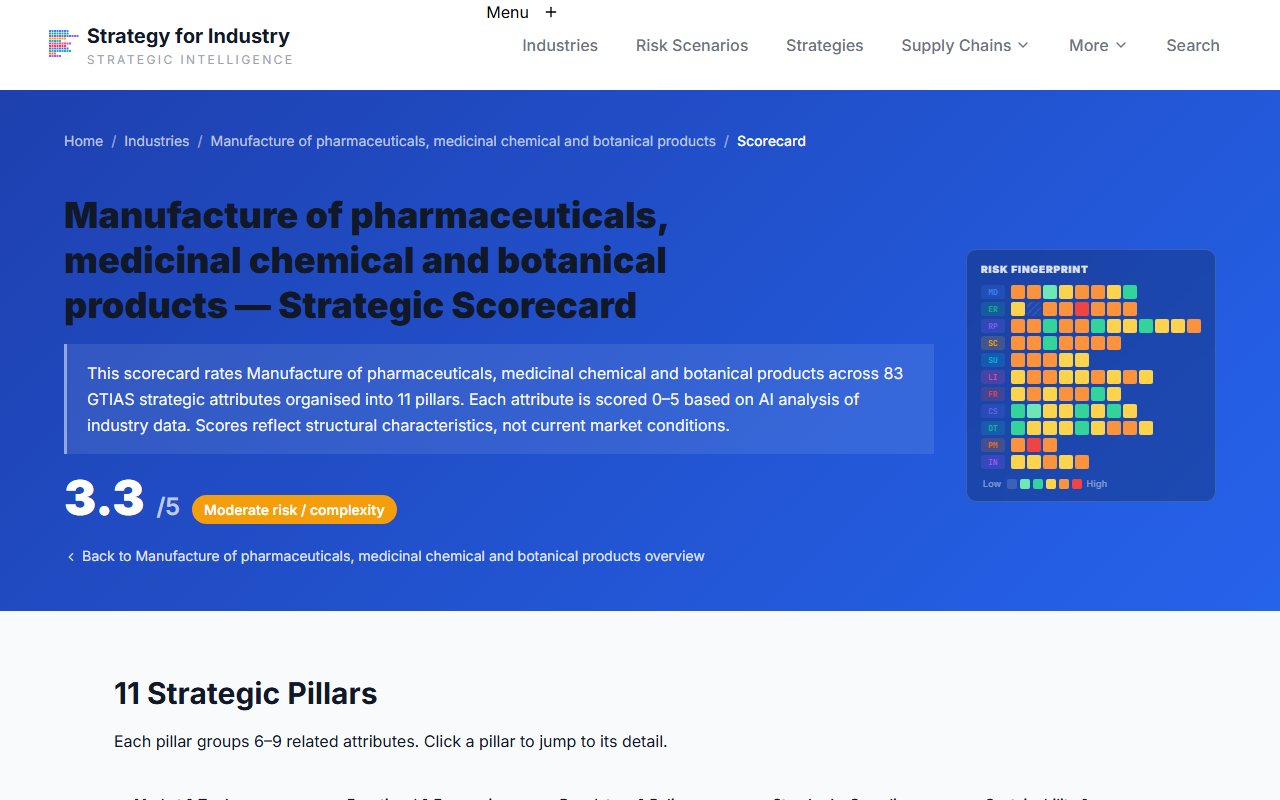
Industry Scorecard
Review the attribute scorecard for trigger evidence
Open the Scorecard page to see the underlying attribute scores driving the confirmed conditions.
Cross-reference the scorecard against the trigger conditions in each matched scenario. Attributes scoring 4–5 that appear in multiple trigger conditions are your highest-priority monitoring targets.
Tactical Playbook
Follow the link to the tactical playbook
From the matched scenario, access the linked playbook for mitigation action steps.
For ESG compliance, look for playbooks in the ESG-ENV and OPS-MFG domains — these will have the supply chain audit and disclosure steps most relevant to your Q3 deadline.
After this journey
You have a structured risk register input: confirmed conditions with trigger evidence, business impact narratives, cascade risk chains, and specific action steps from the playbook — all in one pass without custom research.
Journey 2
Review risk domain exposure across financial, ESG, and digital conditions
Get a domain-level view of where your industry's risk exposure is concentrated before deciding where to focus mitigation resources.
Risk Domains
Go to the Risk Domains index
Open the Risk Domains page to see all domains and their associated risk conditions.
Domains group risk rules by category: FIN (Financial), OPS (Operational), GEO (Geopolitical), ESG (Environmental/Social), DIG (Digital), and HUM (Human). Start here to understand where exposure is concentrated before drilling into specific scenarios.
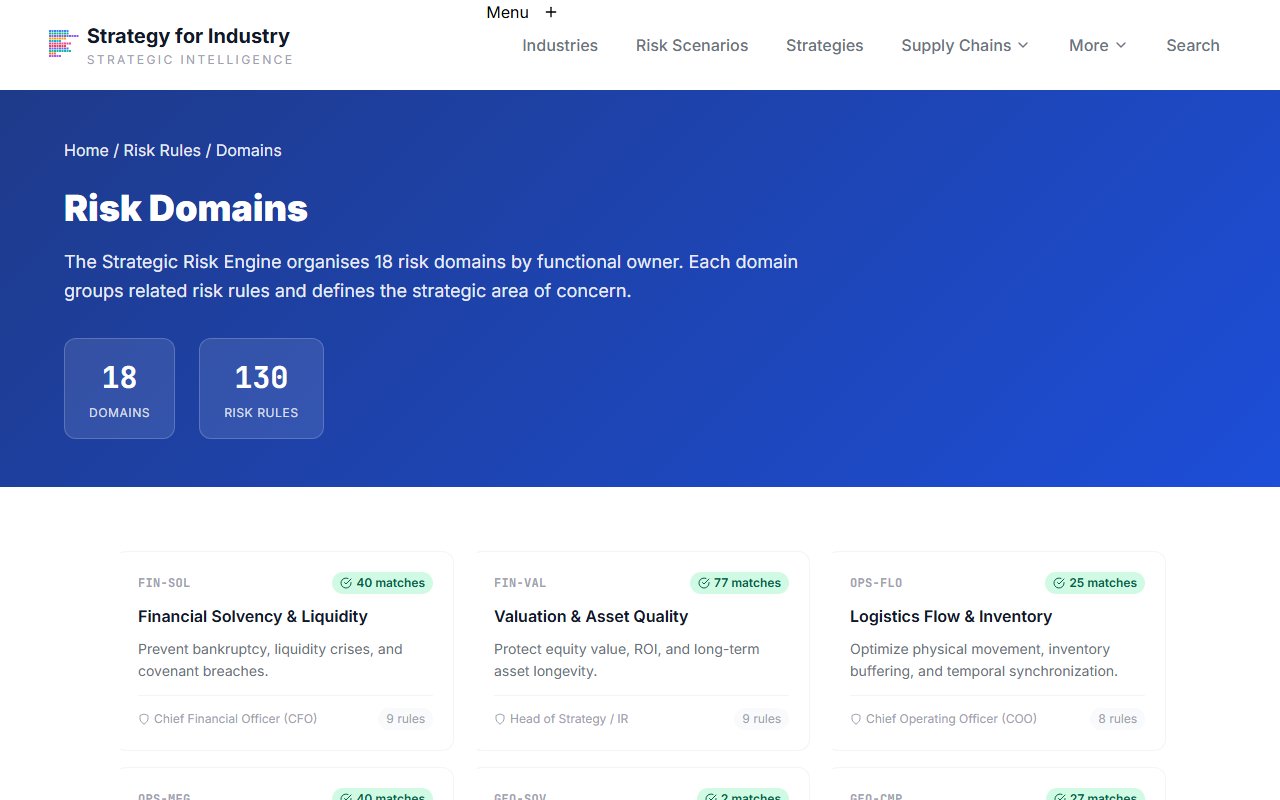
ESG Domain
Open the ESG domain
Click into the ESG-ENV domain for environmental and supply chain ESG conditions.
The ESG-ENV domain contains supply chain due diligence, emissions, and waste management risk rules. For pharmaceutical manufacturing, ESG exposure is high due to hazardous materials handling, packaging waste, and supplier auditing requirements.
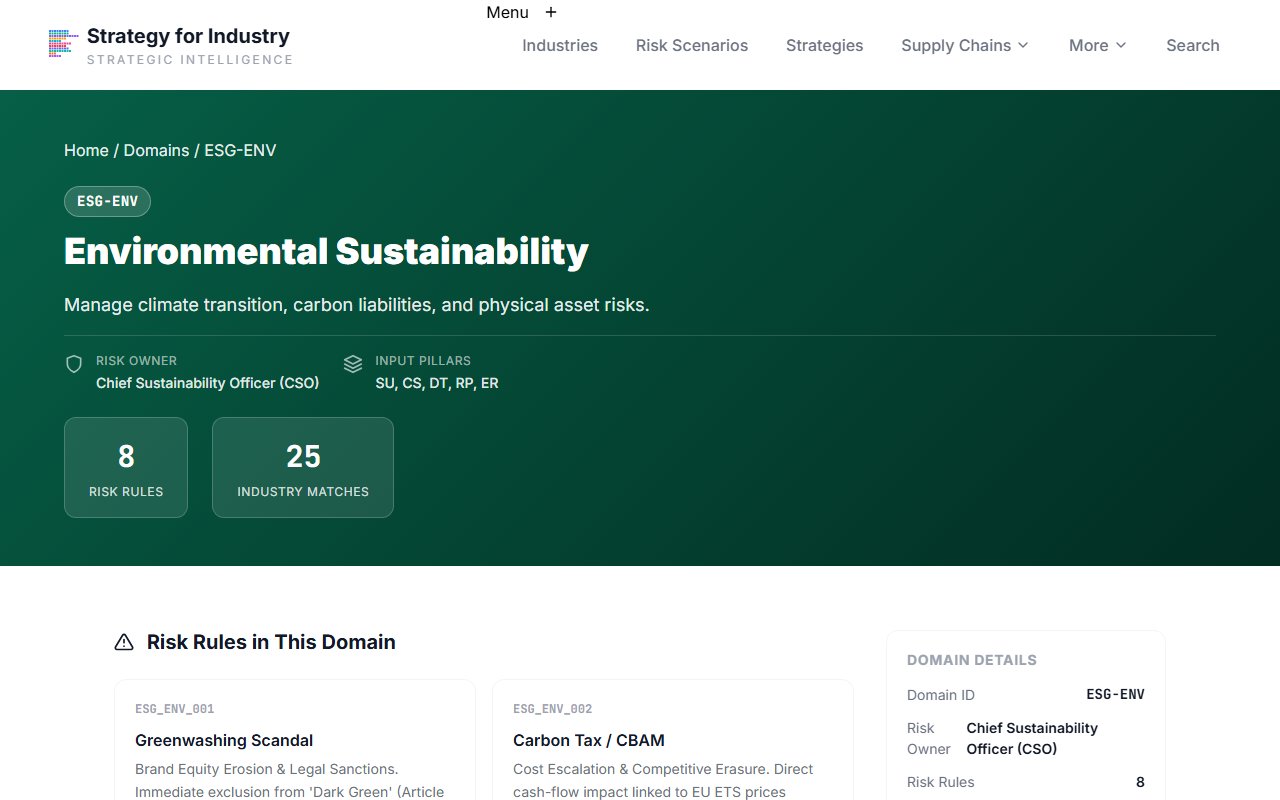
Digital Security Domain
Open the Digital Security domain
Click into the DIG-SEC domain for cybersecurity and digital infrastructure conditions.
Pharmaceutical manufacturing is increasingly digitised — IoT manufacturing, electronic batch records, clinical data systems — and increasingly targeted. DIG-SEC shows whether your industry's digital exposure has triggered any confirmed conditions.
Industry Hub
Cross-reference domain findings with your industry's matched scenarios
Return to your industry hub and compare domain-level findings to confirmed active risks.
Any domain where you found active conditions and your industry has confirmed matches is a priority domain. This gives you a two-level view: sector-wide exposure (domain) plus your specific industry's confirmed triggers (matched scenarios).
After this journey
A domain-level risk heat map for your industry: which domains have the most active triggers, which are systemic to your archetype vs. specific to pharmaceuticals, and where mitigation resources should be concentrated for the Q3 compliance deadline.
Journey 3 — Explore next
Track macro trend exposure for forward-looking risk planning
Go to Macro Trends and review how forces like ESG supply chain due diligence, net-zero decarbonisation, and critical mineral dependencies are projected to amplify existing risk conditions in pharmaceutical manufacturing over the next 1–3 years. Trend-to-industry linkages are pre-mapped with direction (headwind/tailwind) and pressure type.
Browse macro trendsReady to explore?
Start with the working example for this guide — or search for any industry.